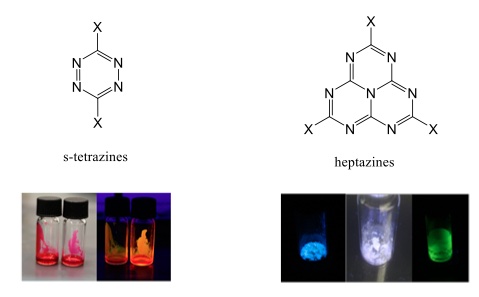
s-Tetrazines1, and the far more enigmatic heptazines2, which count much less described examples, are among the most electron deficient high-nitrogen content, stable aromatic heterocycles (Fig. 1). This peculiarity confers them very original physico-chemical characteristics, including delayed fluorescence, a high electrochemical reduction potential, and a strong potential in organic photocatalysis. In addition, heptazines can trigger enhanced electron transport in OPV devices. However, their synthetic approach, for heptazines, is still in its infancy. We will present and comment on new strategic synthetic procedures involving these two families, insisting more on heptazines recent results, and new properties.
This lecture will therefore recall new synthetic advances in both fields of tetrazines, and heptazines. Noticeably, a new synthetic procedure of heptazines using mechanochemistry, elaborated in the PPSM laboratory, will be presented. As well, we will describe new very low-viscosity tetrazine-based fluorescent liquids. The original delayed fluorescence of original heptazines, which are the first species to present sometimes a singlet-triplet inversion, will be detailed, along with first results in photocatalysis. Examples featuring the molecules concerned are gathered in the Scheme 1 below.
- G. Clavier and P. Audebert “s-Tetrazines as building blocks for new functional molecules and molecular materials”, Chem. Rev., 2010, 110, 3299.
- P. Audebert, E. Kroke, C. Posern and S.-H. Lee, State of art in the preparation, and properties of molecular monomeric s-heptazines: syntheses, characteristics and functional applications Chem. Rev.2021, 121, 2515.